Jan 9, 2018
For years, India’s food safety regulator has twisted rules to allow sale of unsafe processed food
The Food Safety and Standards Authority of India has disregarded the warnings of its own scientists and broken its rules to do so.

By 2025, Indians are expected to eat processed food valued at Rs 72 lakh crore annually, while the revenues of the country’s processed food industry have been forecast to touch Rs 60 lakh crore. To keep growing, India’s food processing industry constantly introduces new flavours and products. Between 2012 and 2015, the food processing industry tried to introduce nearly 4,500 products with completely new formulations.
But, as this two-part series reports, the Indian food safety regime has failed to keep pace with the rapidly growing industry. The first part of the seriesdetails recent Comptroller and Auditor General’s findings on how the authorities allowed the sale of food proven to be dangerous.
In December 2013, the Food Safety and Standards Authority of India, which is responsible for ensuring that the food sold in the country is safe for consumers, granted permission to a company called Pushpam Foods and Beverages to sell an energy drink called Restless Ginseng.
Just under a year later, scientists at the food safety authority gave senior officials a piece of information that is well-recognised in the rest of the world: that caffeine and ginseng, the two key ingredients in Restless Ginseng, make for a dangerous cocktail that could increase heart rate and blood pressure.
It took the authority seven months to react. It was only in June 2015 that it withdrew the no-objection certificate given to the company to sell the product. For a year and a half, the company was able to manufacture and sell a harmful product. Despite the authority’s action, Pushpam Foods continues to promote the drink on its website. The food safety authority has evidently failed to check whether the product has been withdrawn from the market.
As the Comptroller and Auditor General of India pointed out in a recent report, had the authority followed its own guidelines framed in 2011, products such as Restless Ginseng would not have been put in the market in the first place.
The guidelines required food companies to provide scientific evidence that their product is safe. Only after the authority’s scientists were satisfied was the product to be allowed into the market.
Diluting guidelines
Restless Ginseng was not an exception. Starting from 2012, the authority has diluted its regulations, bypassed established protocols and ignored warning from its scientists to allow the sale of more than 800 processed foods with new formulations without assessing their safety. Some of these are still on the market.
The authority enabled this, the CAG noted, by diluting the 2011 guidelines to give temporary one-year no objection certificates to products even before its scientists had examined them.
The dilution goes against the provisions of the Food Safety and Standards Act, 2006, mandating that only scientific panels can decide if a food product or food type is safe to consume.
The bureaucrats heading the authority can issue licences for food business operators to sell a product only after the scientists have approved it as safe.
Proprietary foods
Restless Ginseng is one of hundreds of products that are sold as proprietary or novel foods – new formulations of processed food products for which safety standards do not exist in the 2006 Food Safety and Standards Act that applies to all packaged foods. The 2011 guidelines were formulated to regulate such foods.
Among these foods that do not have pre-set safety standards under the law are energy drinks, salted snacks, noodles, soups and pasta. Also in this category are nutraceuticals – dietary supplements such as multivitamins that may improve health but do not treat a medical condition. Though such supplements might be sold by chemists, they are categorised as food, not medicine.
By 2020, the nutraceutical industry in India alone is expected to grow to Rs 26,000 crore, according to the Drug Marketing and Manufacturing Association.
CAG’s findings
When the CAG did a test audit of 50 proprietary food products approved for sale to consumers between 2012 and 2014, it found that even the diluted regulations were not followed.
In many cases, the authority did not send the product for scientific assessment even after giving it the temporary no-objection certificate. By April 2015, a note written by the authority’s chief executive in May 2015 shows, it had issued such certificates to over a thousand products but sent only 200 of them for testing.
In the case of at least four products, the authority continued to allow sale for up to 47 months after the scientists rejected them as unsafe.
For some products, the authority did not cancel the no-objection certificates in time. Sometimes when it did cancel the certificate, it allowed the sales licence for the product to stand.
The CAG highlighted the case of a company called Chemical International that had received a no-objection certificate to sell a mushroom-based nutraceutical in August 2012. A month later, the authority’s scientists asked for the product to be banned as the company had not submitted clinical data about its claimed health benefits. But the authority did not cancel the licence for the product.
In August 2013, the safety authority gave a company called Surya Herbal a licence to sell Sunova Spirulina tablet. But since the company did not provide scientific evidence of the tablet’s safety, the scientific panel cancelled the no objection certificate in August 2014. Yet, the company continued to carry the licence to sell the tablet till December 2017. It still promotes Spirulina tablets on its website, though it could not be ascertained if this is the same product that has been banned. The authority does not require companies to disclose such details on their websites.
Licence raj
In 27 of the 50 cases audited by the CAG, the authority’s product approval unit had recommended referring the items to its scientific panels for assessment. But this was not done. Instead, the authority issued no-objection certificates to all the products.
Restless Ginseng is probably not the only potentially harmful energy drink that the authority has allowed. The CAG said it was likely other companies besides Pushpam continue to sell drinks with the same dangerous concoction of ginseng and caffeine.
The authority has also granted the approvals arbitrarily. In January 2013, it allowed the Indian biotech company Biocon to market its nutraceutical tablet S-Adenosyl Methionine but denied permission to Sun Pharmaceutical Industries to sell the same product in August that year. Although Biocon’s product approval was withdrawn about a year later, the company continues to hold the licence to sell the nutraceutical till May 2020.
This arbitrary clearance system was struck down when challenged before the Bombay High Court and its decision was validated by the Supreme Court in 2015.
With the clearance regime banned by the courts, the sale of proprietary food products should have stopped until a new system was put in place. But, the CAG found, a month after the judgement of the Bombay High Court, the food safety authority issued “blanket instructions” to its licensing authorities to renew or continue all existing licences issued on the basis of the no-objection certificates it had already issued.
“Consequently, FSSAI permitted the indefinite manufacture, distribution, sale or import of possibly unsafe foods,” the CAG said. “FSSAI did not take any action after the final orders of the Supreme Court to withdraw these blanket instructions.”
Despite this, the food authority, and the Union health and family welfare ministry which oversees it, have been dismissive of the CAG’s report. The main thrust of their defence is that it is old news and a new set of regulations has been put in place starting 2016.
Pawan Kumar Agarwal, the chief executive officer of the Food Safety and Standards Authority of India, reiterated this to Scroll.in. “Product approval [under the guidelines the courts have done away with] is a thing of the past,” he said. “Why don’t you write a forward-looking article? The CAG report is not based on facts.”
But the new regulations promise to be worse, shows an investigation by Scroll.in, which found new evidence that had escaped even the CAG. The details will be reported in the second part of this series.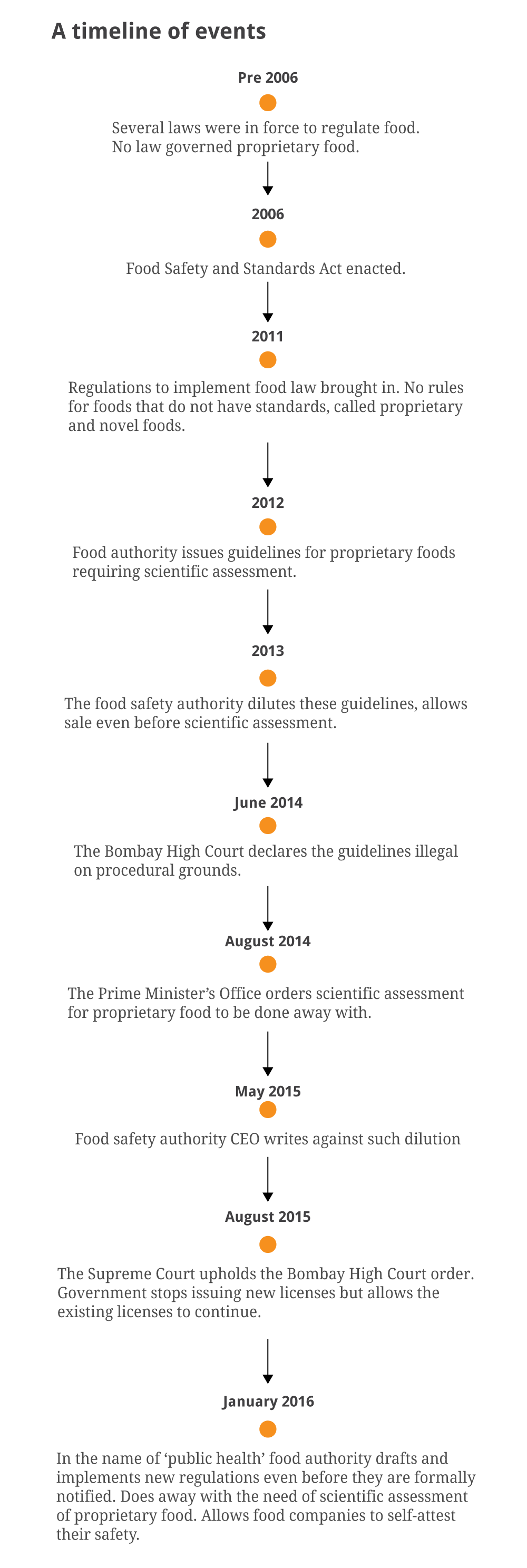

Additives make your food highly unsafe
Herbal products, dietary supplements are risky.
Products containing fluoride and potato protein isolate must be withdrawn.
Hyderabad: The scientific panel of the Food Safety and Standards Authority of India (FSSAI) has recommended that fluoride, potato protein, willow bark extract, lemon balm and pyrroloquinoline quinone (PQQ) must not be used in any food product as they are unsafe for human consumption.
The FSSAI has directed that products containing fluoride and potato protein isolate must be withdrawn immediately and new products must not have these ingredients. Willow bark extract, lemon balm, and PQQ are used in herbal products and dietary supplements and they too are now banned as they are found to have the effect of drugs.
For dietary supplements, functional foods and nutraceuticals these additives are to be used based on the recommended dietary intake guidelines of the FSSAI.
Fluoride
Fluoride is found in foods that contain water. Untreated water has a high fluoride content which is residue from agriculture and industrial waste. The food processing industries that use this water have been told to test the level of fluoride in the water before use. Fluoride is found in liquids more than solids. It is most commonly found in a) Fruit juices; the highest content is found in apple and grape juice b) Sauces like tomato sauce c) Salads that are eaten raw and not properly washed d) Soda e) Toothpaste, mouthwash and dietary supplements.
Excess fluoride in the body causes thyroid, arthritis, dementia, bone fractures and dental fluorosis.
Potato protein
Potato protein isolate is a protein which is extracted from the potato. It has replaced animal protein in the food processing industry to suit vegetarian tastes. It gives a feeling of fullness. Potato proteins are used in a) Baked items like crisp bread, cookies, wafers, crackers, biscuits b) Pizza base, sweet bread, muffins.
High levels of this protein cause kidney problems.
Willow bark
Willow bark is a herb that has been in use since the 5th century. It is used in herbal products, tea and dietary supplements.
The FSSAI says willow bark is nothing but aspirin. Its excessive use in tea powder and herbal products can cause thinning of the blood and in some cases sudden death.
PQQ
Pyrroloquinoline quinine (PQQ) is a compound. It is found naturally in soil and also foods such as kiwi fruit and in human breast milk. It is also added to skimmed milk and dietary supplements.PQQ's role as a vitamin has been controversial since its introduction in 2005. PQQ can be toxic and can damage the kidney and liver. There is a lot of debate about dietary supplements that have PQQ. FSSAI states that it must not be used as its safety parameters have not been identified.
Willow bark
Willow bark is a herb that has been in use since the 5th century. It is used in herbal products, tea and dietary supplements.
The FSSAI says willow bark is nothing but aspirin. Its excessive use in tea powder and herbal products can cause thinning of the blood and in some cases sudden death.
Lemon balm
Lemon balm is a herb of the mint family and has been used for centuries. It is used in herbal teas, ice-creams, ready-to-eat fish foods, health foods and supplements, and mixtures in wines.
There are mixed results for the use of this herb which has good and bad effects. The actual safety dose is difficult to decide as scientists find that it is difficult to quantify. It is found to cause drowsiness, excessive sleep, cognitive decline and also withdrawal symptoms.
FSSAI, Maharashtra FDA and Coca-Cola India join hands to train street food vendors in Pune
Maharashtra’s Food and Drug Administration (FDA) and Coca-Cola India has launched a pilot project in the city to train street food vendors on food safety and good hygiene practices.

This initiative is a part of Memorandum of Understanding (MoU) signed earlier last year by Food Safety & Standards Authority of India (FSSAI) and Coca-Cola to train 50,000 street food vendors across India over next three years under the project ‘Clean Street Food’
This initiative is a part of Memorandum of Understanding (MoU) signed earlier last year by Food Safety & Standards Authority of India (FSSAI) and Coca-Cola to train 50,000 street food vendors across India over next three years under the project ‘Clean Street Food’.
Under this initiative, Coca-Cola and Maharashtra FDA will train 1,200 street food vendors in Pune city in January 2018 on the aspect of safe and nutritious food enabling them to improve the quality of their offering, which will ultimately lead to increase in their earnings.
As part of this initiative, Coca-Cola India, will closely work with FDA in upgrading the eating out experience for the local populace at large. While Coca-Cola India will be responsible for the program execution through its network of trainers and available infrastructure, FDA will ensure the relevance of the training content and shall oversee the overall delivery.
On successful completion of the training, Maharashtra Food and Drug Administration (FDA) will issue a certificate to the street food vendors.
Chief Guest, Girish Bapat, Cabinet Minister for Food, Civil Supplies and Consumer Protection, Food and Drugs Administration and Parliamentary Affairs, present at the launch said, ”Street Food Vendors form an integral part of our society and reflect the local food culture. While Street Food Vendors offer nutritional and affordable food to consumers, it would be highly beneficial for them to upgrade their knowledge of food safety standards and rules of hygiene. This joint initiative between FDA and Coca-Cola India is a positive step towards promoting safe and nutritious food while eating out.”
Commenting on the pilot, Pallavi Darade, Commissioner Maharashtra FDA said, “Under this program launched jointly with Coca-Cola India, we have selected Pune as the first city to launch the pilot initiative due to its vibrant and diverse food culture. Our collaboration with Coca-Cola India will help us in sensitizing street vendors on practising good hygiene and maintain cleanliness to create a good eating out experience for consumers.”
Darade further mentioned that under an earlier project ‘Bhog’ her team had raised the standards of ‘traditional Prasad’ being served in temples across the state. The world famous ‘Shree Siddhivinayak’ temple in Mumbai was the first one to adopt these initiatives and subsequently was cleared by USFDA to allow dispatch of ‘prasad’ to devotees in the United States.
The training under Clean Street Food program is delivered through a unique classroom on wheels. This customized bus is fully equipped with training pre-requisites such as a seating area, presentation zone, audio-video equipment and on-board trainers will reach out to street vendors in rural and semi-urban areas with the help of local FDA teams.
Speaking on the occasion, Ishteyaque Amjad, Vice President Public Affairs and Communication, Coca-Cola India and South West Asia said, “Our collaboration with FSSAI and various State FDAs for project ‘Clean Street Food’ is aimed at providing best practises and knowledge to street food vendors through specialised training modules enabling them grow their businesses sustainably. The initiative has been designed by keeping in mind the specific needs of the small food vendors. Coca-Cola India is committed to build capabilities and support sustainable food security programs.”
Over the next three years, the Clean Street Food project will expand to 14 other locations across nine different states through the bottling plant network of Coca-Cola India states including Uttar Pradesh, Maharashtra, Andhra Pradesh, West Bengal, Chandigarh, Punjab, Chhattisgarh, Delhi NCR.
‘Plastic’ alert in a few batches of Danish chocolate
Summary: Pune: The national food regulatory body has issued an alert over some batches of Danish chocolates imported by India which may cause choking in children. The European Commission sounded the alert first about plastic fragments in the chocolates.The chocolates in question are Anthon Berg Marcipanbrod Whiskey 40g, Anthon Berg Marcipanbrod Cognac 40g, Anthon Berg Sweet Moments Marzipan Bar Mini 165g and Anthon Berg Sweet Moments Marzipan Collection 400g. Our officials have been told to visit malls and other shops that sell imported chocolates. "There is a risk that the product in question contains small pieces of plastic which may cause choking in children. We are keeping an eye over the online sales.
Pune: The national food regulatory body has issued an alert over some batches of Danish chocolates imported by India which may cause choking in children. The European Commission sounded the alert first about plastic fragments in the chocolates.The chocolates in question are Anthon Berg Marcipanbrod Whiskey 40g, Anthon Berg Marcipanbrod Cognac 40g, Anthon Berg Sweet Moments Marzipan Bar Mini 165g and Anthon Berg Sweet Moments Marzipan Collection 400g."There is a risk that the product in question contains small pieces of plastic which may cause choking in children. It is imported by India and hence imperative to start follow-up action, including recall," P Kartikeyan, assistant director (regulations), Food Safety and Standards Authority of India said in the alert notification issued on Friday.The product category under which the chocolates are sold is cocoa and cocoa preparations, coffee and tea , As Reported By TOI.
According to the Newspaper,They are likely to reach consumers through online sales or by other means.Chandrashekhar Salunkhe, joint commissioner (HQ), state Food and Drug Administration (FDA) said, " We have started tracking the importers of these specific chocolates. Our officials have been told to visit malls and other shops that sell imported chocolates. We are keeping an eye over the online sales."The European Commission, through an alert, has informed Belarus, Belgium, Canada, Finland, France, Germany, Hungary, Norway, Qatar, Slovakia, Sweden , Turkey and the United Kingdom about the product and follow-up has commenced, according to the Union ministry of health and family welfare.When contacted, an official from Dorabjee's supermarket and grocery store in Camp area said they did not stock these Danish chocolates.
Subscribe to:
Posts (Atom)


